Hip fractures are estimated to affect 500,000 adults in the United States each year. They have a tremendous impact on quality of life, currently accounting for 3-7% of in-hospital mortality and a one-year mortality rate of 19.4-58%. Approximately half of all hip fractures are intertrochanteric (IT) fractures. The two most common implants used to treat IT fractures are the cephalomedullary nail (CMN) and the sliding hip screw (SHS).
Despite advances in technology, bone discontinuity and fixation failures such as penetration of the femoral head still occur and can be devastating. Recent studies have reported resection rates of up to 6% with modern implants. Risk factors for fixation failure include tip-tip distance (TAD) >25 mm, inadequate fracture repositioning, unstable fracture patterns, cephalic spikes located to the side or below the center of the femoral head, and internal rotation of the cervical stem angle. Aging and osteoporosis are also associated with fixation failure.
Patients and Methods
This was a retrospective survey of patients treated at two institutions located within a healthcare system in a large metropolitan area. The case logs of two surgeons (JS and BC) were queried for all patients who received cement-reinforced revision fixation after failure of initial fixation of IT fractures from January 2018 to September 2021, after approval by the institutional review board. The use of cement reinforcement during revision surgery was confirmed by electronic medical record (EMR) review of operative records and postoperative radiographs. We included all patients with signs of cemented strengthening in revision fixation after failed initial CMN or SHS fixation of IT fractures (Figure 1). We excluded patients with femoral neck or subtrochanteric fractures, patients who did not undergo revision fixation with cement reinforcement, and patients who underwent arthroplasty at the time of the initial revision surgery.
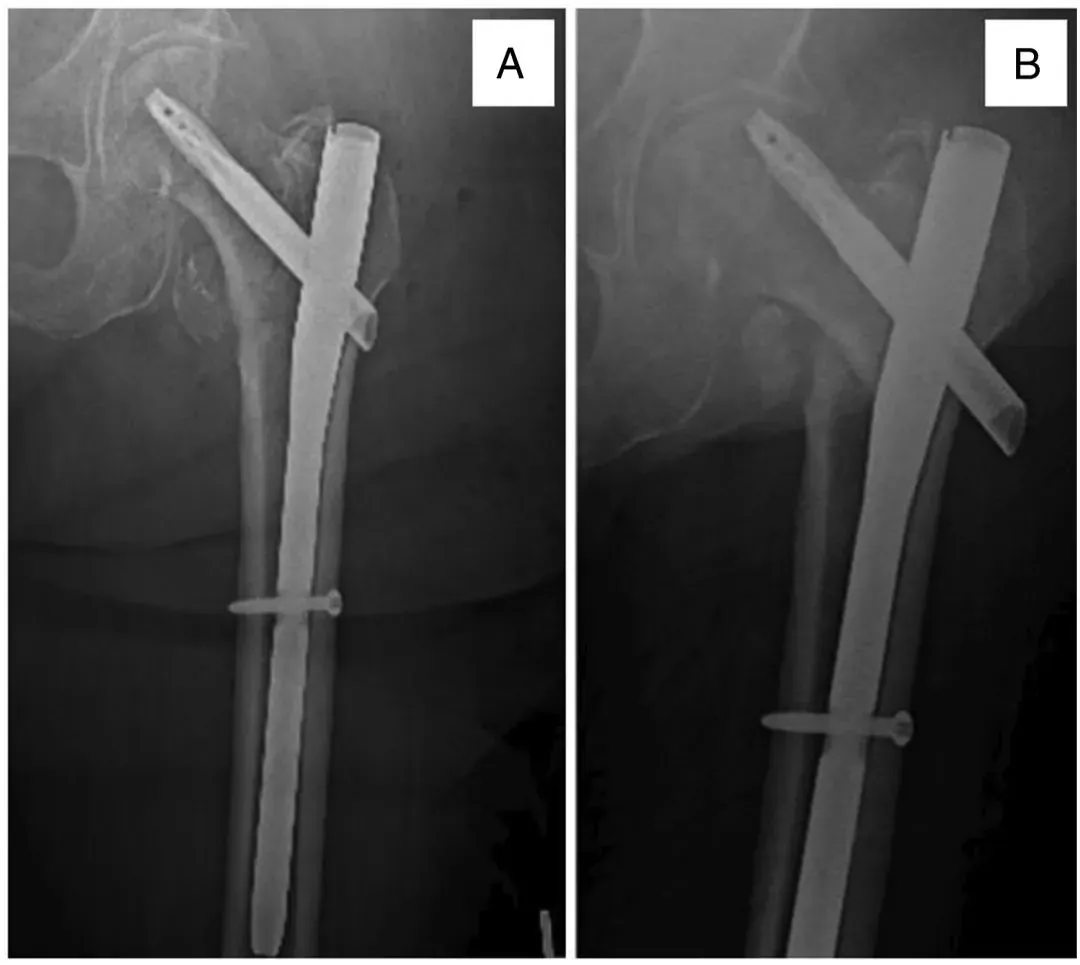
Figure 1. A 76-year-old woman underwent intramedullary nailing for an intertrochanteric fracture of the femur (A), with persistent hip pain 2 months later due to the implant incision (B).
Surgical techniques
For patients receiving initial fixation with CMN, we performed a modified head nail replacement and bone cement reinforcement. Initially, a 5-cm incision was made on the proximal side of the greater trochanter, a guide wire was placed into the proximal side of the nail, and all bone was removed from the proximal side of the nail using an open reamer. Next, a hexagonal screwdriver was used to loosen the fixation screw at the top of the CMN (Figure 2).
![Set screw]()
Figure 2, intraoperative fluoroscopy showing a hexagonal screwdriver engaging and loosening a previously nailed set screw.
A 1-2 cm transverse incision is then made down through the iliotibial fascia (ITB) fascia. The original head nail is removed with a reverse threaded guide (Figure 3). In the case of femoral head perforation, a hollow guide with a distal locking screw is used in order to prevent leakage of cement into the joint (Figure 3A). Specifically, the matrix was first injected after removal of the outer two layers of the triple cannula and then put into the perforated area by reinserting the outer two layers.
![Intramedullary nail]()
Figure 3, Intraoperative anteroposterior (A) and lateral (B) images of another patient showing femoral head with interstitial bone defect graft.
Next, the limb is tractioned and then the fracture is repositioned into more of an exostosis for revision fixation. In cases of malunion or fibrous healing, a percutaneous osteotomy is performed using a 1/4" bone gouge with an anterolateral approach. This is rarely necessary, but very effective when needed to produce an improved cervical stem angle (target >130°).
A new screw or spiral blade is then placed into the subchondral bone in the femoral head along the axis of the femoral neck, making sure to be careful not to penetrate the head (Figure 4). The screw is placed intentionally avoiding the previous nail tract, but still pointing to end at the center of the femoral head. (Figure 5)
![Intramedullary nail]()
Figure 4, Anteroposterior (A) and lateral (B) images of another patient showing the insertion of the kerfing needle along the path of the new head nail.
![Intramedullary nail]()
Figure 5, intraoperative fluoroscopy showing the insertion of a new cephalomedullary blade along the guide wire path, which was subsequently tightened by a set screw.
Finally, the femoral head is filled with bone cement using an injectable bone cement system (Figure 6). Care is taken to avoid extrusion into the joint by using real-time radiographs and adjusting the depth and orientation of the cement cannula.
![Intramedullary nail]()
Figure 6, Imaging showing cement increase initially (A), gradual filling (B) until the femoral head defect is filled (C).
For patients receiving initial fixation of the SHS, we remove the SHS and place a long CMN. after making a 5-cm incision centrally below the greater trochanter and identifying the ITB, the incision is dissected down to the lateral plate. All plate screws are removed using a suitable hand screwdriver and the lateral plate is then removed. The tension screws are then removed with a reverse thread guide as previously described to reposition the fracture to a greater degree of valgus. A 5-cm incision is then made at the tip of the greater trochanter as previously described. A guide wire is inserted at the most proximal end of the greater trochanter and advanced into the femoral stem. An open reamer is introduced along the path of the guidewire. A long ball-tipped guidewire is then threaded into the center of the distal femur below the level of the patella. Next, progressive reaming was performed until tremor was felt in the backbone. All of our patients received a long CMN nail with the TFN-ADVANCED (TFNa) proximal femoral intramedullary nailing system (DePuy-Synthes, Raynham, MA).
Discussion
Our technique uses bone cement reinforcement during revision fixation. Bone cement reinforcement for initial fixation of osteoporotic proximal femur fractures has been studied and has shown good biomechanical and clinical results. A recent review reported that bone cement reinforcement resulted in higher failure loads, reduced implant displacement, and fewer complications and reoperations compared to nonreinforced fixation. A randomized, multicenter prospective study also reported no reoperations or symptomatic episodes of CMN displacement in the cement-reinforced group compared with six cases in the nonreinforced group.
Hip fracture mortality is high. Recent studies have described a 3-7% in-hospital mortality rate, with one-year mortality decreasing from 19.4% to 58%. In particular, IT fractures have been shown to account for 27% of mortality at one year. Our clinical series showed no in-hospital mortality and a one-year mortality rate of 13.6%, which is relatively low compared to the literature. Because early bed mobility after surgery is associated with lower mortality, good ambulation and functional outcomes in our series could explain the relatively low mortality observed in our patients.
Conclusion
Cement-reinforced revision fixation is an effective, safe, and cost-effective method for non-infected intertrochanteric fracture fixation failure in elderly patients with adequate acetabular bone stock.
Patients with failed fixation of primary intertrochanteric fractures followed by revision fixation and cement-reinforced treatment have demonstrated good long-term clinical and quality-of-life outcomes. The procedure was used only in cases where the majority of the acetabular articular surface was preserved and the head nail remained contained within the femoral neck. Considering the limitations of revision arthroplasty in frailer elderly patients, this procedure shows promise in safely and effectively reducing serious complications while reducing operative time and cost in this group of patients.
How to Buy Orthopaedic Implants and Orthopaedic Instruments?
For CZMEDITECH, we have a very complete product line of orthopedic surgery implants and corresponding instruments, the products including spine implants, intramedullary nails, trauma plate, locking plate, cranial-maxillofacial, prosthesis, power tools, external fixators, arthroscopy, veterinary care and their supporting instrument sets.
In addition, we are committed to continuously developing new products and expanding product lines, so as to meet the surgical needs of more doctors and patients, and also make our company more competitive in the whole global orthopedic implants and instruments industry.
We export worldwide, so you can contact us at email address song@orthopedic-china.com for a free quote, or send a message on WhatsApp for a quick response +86-18112515727.
If want to know more information,click CZMEDITECH to find more details.
Español
Русский
Français
English
العربية
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu
















