INTRODUCTION
Global Intramedullary Nail Market Overview
As orthopedic trauma procedures continue to increase worldwide, hospitals, distributors, and procurement teams are placing greater emphasis on supplier stability, product consistency, and long-term cooperation capability.
In the intramedullary nail market, purchasing decisions are no longer based only on pricing. More distributors are evaluating manufacturers based on:
Complete trauma product portfolios
OEM/private label flexibility
Instrument compatibility
Regulatory compliance
Delivery reliability
International market experience
For orthopedic distributors, selecting the right manufacturing partner can directly influence market competitiveness, hospital cooperation opportunities, and procurement efficiency.
This article highlights internationally recognized intramedullary nail manufacturers commonly referenced by orthopedic distributors and hospital buyers worldwide.
How Global Distributors Choose Orthopedic Trauma Suppliers
For orthopedic distributors and hospital procurement teams, selecting an intramedullary nail manufacturer is a strategic sourcing decision.
The right supplier can improve:
The following factors are among the most important when evaluating orthopedic trauma manufacturers.
Complete Orthopedic Trauma Product Portfolio
Many distributors prefer suppliers that can provide:
Working with a complete orthopedic supplier helps reduce sourcing complexity and improves purchasing efficiency.
OEM / Private Label Support
OEM and private label capability have become increasingly important for orthopedic distributors building regional brands.
Key OEM requirements include:
Regulatory Compliance
International orthopedic distribution requires reliable regulatory support.
Most buyers prioritize suppliers with:
Instrument Compatibility & Surgical Support
Hospitals and surgeons require stable instrument compatibility for trauma procedures.
Distributors often prioritize suppliers offering:
Complete targeting device systems
Compatible trauma instruments
Reusable instrument trays
Stable surgical system integration
Orthopedic Portfolio Considerations
Many distributors are gradually moving toward integrated trauma sourcing strategies rather than purchasing from multiple isolated suppliers.
Manufacturers capable of supporting broader orthopedic product categories may help distributors improve sourcing efficiency and long-term portfolio planning.
Explore Orthopedic Trauma Product Portfolio Options
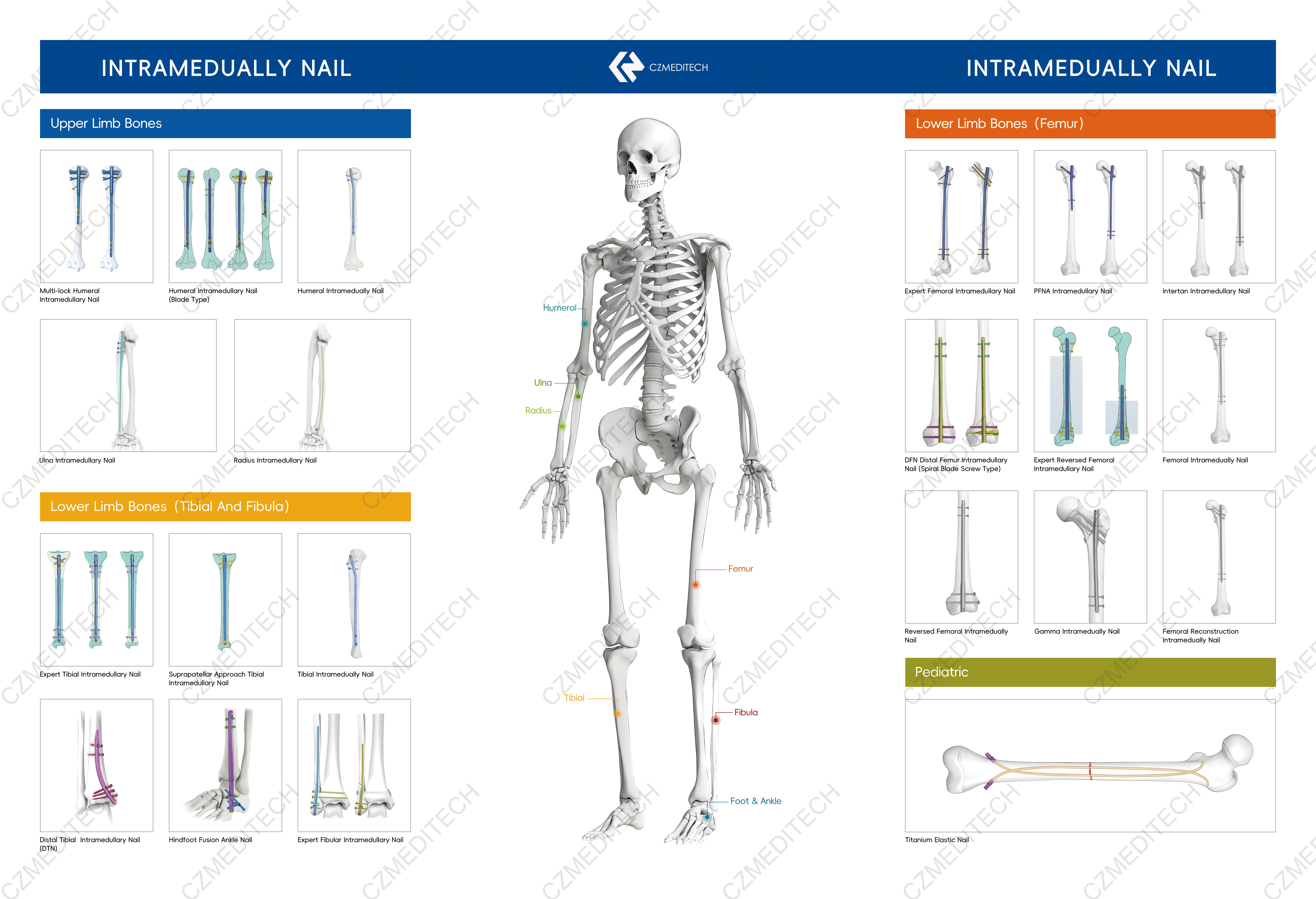
Top 25 Intramedullary Nail Manufacturers and Suppliers
1. Stryker (USA)
Company Overview
Stryker is one of the world’s largest orthopedic medical technology companies specializing in trauma, reconstruction, navigation, and surgical systems.
Main Intramedullary Nail Products
Strengths
Strong global hospital network
Advanced surgical technologies
Premium orthopedic positioning
Strong surgeon recognition
Suitable Buyers
Large hospitals, government tenders, premium orthopedic distributors.
2. Zimmer Biomet (USA)
Company Overview
Zimmer Biomet is a globally recognized orthopedic implant manufacturer with extensive experience in trauma and reconstruction systems.
Main Products
Strengths
Strong clinical recognition
Global distribution network
Advanced orthopedic technologies
Suitable Buyers
Hospital procurement groups and established orthopedic distributors.
3. CZMEDITECH (China)
Company Overview
CZMEDITECH is a China-based orthopedic implant and instrument manufacturer serving orthopedic distributors, hospitals, surgical centers, and OEM/ODM partners worldwide.
The company specializes in:
Intramedullary nail systems
Locking plate systems
External fixation systems
Spine implants
Sports medicine products
Orthopedic power tools
Surgical instrument systems
CZMEDITECH focuses on helping distributors build complete orthopedic trauma product portfolios with flexible cooperation models and stable manufacturing support.
Main Intramedullary Nail Products
Strengths
✔ Complete orthopedic trauma product portfolio
✔ Strong OEM/ODM manufacturing capability
✔ Distributor-friendly cooperation model
✔ Competitive global pricing
✔ Flexible MOQ support
✔ Fast production and delivery
✔ CE / ISO documentation support
✔ Tender project assistance
Suitable Buyers
Orthopedic distributors
OEM/private label buyers
Hospital procurement teams
Government tender suppliers
Emerging market distributors
Why CZMEDITECH Is Suitable for Orthopedic Distributors
Unlike suppliers focused only on trauma nails, CZMEDITECH can provide integrated orthopedic sourcing solutions including:
Intramedullary nails
Locking plates
External fixators
Trauma screws
Spine systems
Orthopedic instruments
Power tools
This allows distributors to reduce sourcing complexity and expand their orthopedic product portfolios through one supplier partnership.
Request Orthopedic Product Portfolio Information
4. Smith & Nephew (UK)
Company Overview
Smith & Nephew is an international medical technology company specializing in trauma reconstruction and sports medicine solutions.
Strengths
Innovation-driven orthopedic technologies and strong surgeon education support.
Suitable Buyers
Premium hospitals and orthopedic surgical centers.
5. B. Braun / Aesculap (Germany)
Company Overview
B. Braun Aesculap is known for precision-engineered orthopedic systems and strong European clinical positioning.
Strengths
High manufacturing standards and strong hospital recognition within Europe.
Suitable Buyers
European hospitals and premium distributors.
6. Globus Medical (USA)
Company Overview
Globus Medical is a fast-growing orthopedic company specializing in spine and trauma technologies with expanding international distribution capability.
Strengths
Strong innovation capability, expanding trauma product portfolio, and growing global market presence.
Suitable Buyers
Modern orthopedic centers, spine-focused distributors, and trauma implant procurement teams.
7. Acumed (USA)
Company Overview
Acumed is an orthopedic trauma company specializing in upper and lower extremity fixation systems and trauma solutions.
Strengths
Strong extremity trauma specialization and surgeon-focused product development.
Suitable Buyers
Orthopedic trauma specialists, hospitals, and specialty distributors.
8. Arthrex (USA)
Company Overview
Arthrex is an orthopedic technology company focused on minimally invasive surgical systems and sports medicine innovation.
Strengths
Advanced surgical technologies, strong surgeon education programs, and global sports medicine recognition.
Suitable Buyers
Sports medicine centers, premium hospitals, and orthopedic surgical distributors.
9. Citieffe (Italy)
Company Overview
Citieffe is a European orthopedic implant manufacturer specializing in trauma fixation systems and intramedullary nail technologies.
Strengths
Strong trauma product specialization and established European distribution experience.
Suitable Buyers
European hospitals, orthopedic distributors, and trauma procurement groups.
10. Medtronic Trauma Division (USA)
Company Overview
Medtronic Trauma Division is part of the global medical technology company Medtronic, providing diversified orthopedic trauma solutions.
Strengths
Strong international regulatory capability, hospital integration experience, and global healthcare presence.
Suitable Buyers
Large hospital systems, government procurement projects, and international healthcare networks.
11. Wright Medical (Stryker Group)
Company Overview
Wright Medical is an orthopedic extremity and trauma specialist integrated into Stryker’s global orthopedic business network.
Strengths
Strong extremity orthopedic technologies and access to Stryker’s international distribution resources.
Suitable Buyers
Premium orthopedic hospitals, surgical centers, and specialty trauma distributors.
12. Orthofix (USA)
Company Overview
Orthofix is an orthopedic fixation company specializing in trauma implants, bone healing technologies, and orthopedic surgical solutions.
Strengths
Strong trauma fixation expertise and advanced bone healing technologies.
Suitable Buyers
Trauma centers, orthopedic hospitals, and specialized trauma distributors.
13. Tecomet Orthopedics (USA)
Company Overview
Tecomet Orthopedics is an OEM-focused orthopedic manufacturing supplier providing precision implant manufacturing solutions.
Strengths
Strong OEM manufacturing capability, precision engineering, and private label production support.
Suitable Buyers
OEM brands, private label distributors, and orthopedic manufacturing partners.
14. Sanatmetal (Hungary)
Company Overview
Sanatmetal is a European orthopedic implant manufacturer with international export and OEM cooperation experience.
Strengths
European manufacturing quality, flexible cooperation models, and established export capability.
Suitable Buyers
European distributors, emerging orthopedic markets, and OEM partners.
15. Auxein Medical (India)
Company Overview
Auxein Medical is an orthopedic trauma implant exporter serving international orthopedic distribution markets.
Strengths
Competitive manufacturing costs, export-oriented operations, and broad trauma implant coverage.
Suitable Buyers
Emerging market distributors, regional importers, and cost-sensitive procurement projects.
16. GPC Medical (India)
Company Overview
GPC Medical is a cost-effective orthopedic implant manufacturer with long-term international export experience.
Strengths
Flexible production capability, competitive pricing, and OEM cooperation support.
Suitable Buyers
Regional orthopedic distributors, Africa and Asia markets, and private label buyers.
17. Response Ortho (Turkey)
Company Overview
Response Ortho is an orthopedic trauma manufacturer serving Europe, the Middle East, and regional distribution markets.
Strengths
Strategic regional logistics advantages and competitive trauma implant manufacturing capability.
Suitable Buyers
European distributors, Middle East importers, and regional orthopedic suppliers.
18. DePuy Synthes (USA)
Company Overview
DePuy Synthes, part of Johnson & Johnson, is a globally recognized orthopedic trauma company with strong hospital market presence.
Strengths
Global orthopedic brand recognition, advanced trauma technologies, and extensive surgeon network support.
Suitable Buyers
Large hospitals, premium orthopedic distributors, and government procurement projects.
19. NuVasive (USA)
Company Overview
NuVasive is an orthopedic technology company specializing in spine and trauma surgical solutions.
Strengths
Innovation-driven surgical technologies and strong spine surgery market positioning.
Suitable Buyers
Spine-focused hospitals, surgical centers, and orthopedic technology distributors.
20. Exactech (USA)
Company Overview
Exactech is an orthopedic implant manufacturer serving trauma reconstruction and orthopedic surgery markets.
Strengths
Strong orthopedic product development capability and surgeon-oriented solutions.
Suitable Buyers
Orthopedic hospitals, reconstruction centers, and regional distributors.
21. Bioimpianti (Italy)
Company Overview
Bioimpianti is a European orthopedic implant company with trauma fixation and orthopedic reconstruction product lines.
Strengths
European orthopedic manufacturing experience and trauma fixation specialization.
Suitable Buyers
European distributors, trauma hospitals, and orthopedic procurement groups.
22. SIGN Fracture Care International
Company Overview
SIGN Fracture Care International is an orthopedic organization and implant provider focusing on improving trauma care accessibility globally.
Strengths
Global trauma care support programs and focus on underserved healthcare markets.
Suitable Buyers
Humanitarian healthcare organizations, developing healthcare systems, and trauma support programs.
23. Aap Implantate (Germany)
Company Overview
Aap Implantate is a German orthopedic implant manufacturer specializing in trauma fixation technologies.
Strengths
German engineering standards and trauma fixation product specialization.
Suitable Buyers
European hospitals, trauma distributors, and orthopedic procurement teams.
24. Newclip Technics (France)
Company Overview
Newclip Technics is an orthopedic implant company focusing on trauma and extremity fixation technologies.
Strengths
Strong extremity fixation expertise and innovative orthopedic product development.
Suitable Buyers
Extremity trauma specialists, orthopedic centers, and European distributors.
25. TST Medical
Company Overview
TST Medical is an OEM-oriented orthopedic implant supplier serving international orthopedic distribution markets.
Strengths
Flexible OEM cooperation capability and export-oriented orthopedic manufacturing support.
Suitable Buyers
Private label distributors, OEM partners, and regional orthopedic importers.
Evaluating Orthopedic Trauma Suppliers for Your Market?
Distributors often compare:
CZMEDITECH supports global distributors with integrated orthopedic trauma solutions and OEM cooperation support.
Request Product Portfolio Information
Manufacturer Comparison Table
| Rank | Company | Country | OEM / ODM Capability | Trauma Product Portfolio | Instrument Support | Market Position | Suitable Buyers |
| 1 | Stryker | USA | Limited | Complete Trauma Portfolio | Strong | Premium Global Brand | Large Hospitals / Government Tenders |
| 2 | Zimmer Biomet | USA | Limited | Complete Orthopedic Portfolio | Strong | Premium Hospital Market | Hospital Networks / Procurement Groups |
| 3 | CZMEDITECH | China | Strong | Integrated Trauma Portfolio | Strong | Distributor & OEM Focused | Distributors / OEM Partners |
| 4 | Smith & Nephew | UK | Limited | Trauma & Sports Medicine | Strong | Premium Surgical Market | Surgical Centers / Hospitals |
| 5 | B. Braun / Aesculap | Germany | Limited | Trauma-Focused Portfolio | Strong | European High-end Market | EU Hospitals / Premium Buyers |
| 6 | Globus Medical | USA | Partial | Spine & Trauma Portfolio | Moderate | Innovation-driven Brand | Spine Centers / Trauma Buyers |
| 7 | Acumed | USA | Limited | Extremity Trauma Systems | Moderate | Specialist Trauma Brand | Orthopedic Specialists |
| 8 | Arthrex | USA | Limited | Sports Medicine & Trauma | Strong | Premium Surgical Brand | Sports Medicine Centers |
| 9 | Citieffe | Italy | Partial | Trauma & Fixation Systems | Moderate | European Trauma Supplier | EU Distributors / Hospitals |
| 10 | Medtronic Trauma Division | USA | Limited | Diversified Orthopedic Systems | Strong | Global Medical Technology Brand | Healthcare Systems |
| 11 | Wright Medical (Stryker Group) | USA | Limited | Extremity & Trauma Systems | Strong | Premium Orthopedic Market | Orthopedic Hospitals |
| 12 | Orthofix | USA | Partial | Trauma & Bone Healing | Strong | Trauma Technology Brand | Trauma Centers |
| 13 | Tecomet Orthopedics | USA | Strong | OEM Manufacturing Focus | OEM-based | Precision Manufacturing Supplier | Private Label Brands |
| 14 | Sanatmetal | Hungary | Strong | Trauma Implant Portfolio | Moderate | European OEM Supplier | EU & Emerging Markets |
| 15 | Auxein Medical | India | Strong | Trauma Implant Systems | Moderate | Export-focused Manufacturer | Emerging Market Distributors |
| 16 | GPC Medical | India | Strong | Orthopedic Trauma Portfolio | Moderate | Cost-effective Supplier | Africa / Asia Importers |
| 17 | Response Ortho | Turkey | Strong | Trauma & Fixation Systems | Moderate | Regional Export Supplier | Middle East / EU Buyers |
| 18 | DePuy Synthes | USA | Limited | Complete Trauma Portfolio | Strong | Premium Global Orthopedic Brand | Large Hospitals |
| 19 | NuVasive | USA | Partial | Spine & Trauma Systems | Moderate | Technology-focused Brand | Spine-focused Buyers |
| 20 | Exactech | USA | Partial | Reconstruction & Trauma | Moderate | Orthopedic Surgical Brand | Orthopedic Centers |
| 21 | Bioimpianti | Italy | Partial | Trauma & Reconstruction | Moderate | European Orthopedic Supplier | EU Distributors |
| 22 | SIGN Fracture Care International | International | Limited | Trauma Care Solutions | Moderate | Nonprofit Trauma Organization | Developing Healthcare Markets |
| 23 | Aap Implantate | Germany | Partial | Trauma Fixation Systems | Moderate | German Trauma Implant Brand | EU Trauma Buyers |
| 24 | Newclip Technics | France | Partial | Extremity & Trauma Systems | Moderate | Extremity-focused Brand | Specialty Orthopedic Centers |
| 25 | TST Medical | China | Strong | OEM Trauma Manufacturing | Moderate | OEM-oriented Supplier | Private Label Distributors |
Supplier Selection for Different Market Needs
| Procurement Requirement | Recommended Manufacturer |
| OEM / Private Label Cooperation | CZMEDITECH |
| Integrated Trauma Portfolio | CZMEDITECH |
| Premium Hospital Market | Stryker |
| European Hospital Market | B. Braun |
| Government Tender Projects | Zimmer Biomet |
| Trauma Center Specialization | Orthofix |
| Surgical Innovation Focus | Smith & Nephew |
Why More Distributors Prefer Integrated Orthopedic Suppliers
Many orthopedic distributors are gradually shifting from single-category purchasing models toward integrated orthopedic sourcing strategies.
Instead of sourcing implants, instruments, and trauma systems separately, distributors increasingly prefer manufacturers capable of supporting:
Multi-category trauma portfolios
Instrument compatibility
Consistent supply coordination
OEM/private label cooperation
Long-term distributor support
This approach may help distributors improve procurement efficiency while simplifying supplier management.
Among manufacturers serving distributor-oriented cooperation models, integrated orthopedic product capability is becoming an increasingly important factor in supplier selection.
Review Available Orthopedic Trauma Categories
OEM & Distribution Cooperation
For distributors exploring OEM or private label orthopedic cooperation, manufacturers typically differ in:
Product category breadth
Production flexibility
Documentation support
Branding capability
Export experience
CZMEDITECH supports distributor-oriented cooperation models including:
Product portfolio coordination
OEM/private label support
Instrument system integration
Export documentation assistance
Distributor cooperation discussions
Request Product Portfolio & Cooperation Information
Related Orthopedic Trauma Categories
FAQ
Which intramedullary nail manufacturers support OEM cooperation?
Some orthopedic manufacturers provide OEM/private label cooperation for distributors and regional orthopedic brands.
What factors are important when selecting an orthopedic trauma supplier?
Common evaluation factors include product quality consistency, trauma portfolio completeness, OEM flexibility, documentation support, and delivery stability.
Why do distributors prefer integrated orthopedic suppliers?
Integrated orthopedic sourcing may help reduce procurement complexity and improve product coordination across trauma categories.
What certifications are commonly required for orthopedic implant distribution?
CE certification and ISO 13485 quality management systems are commonly required in international orthopedic markets.
CONCLUSION
The global intramedullary nail market continues to evolve as orthopedic distributors and hospital procurement teams increasingly prioritize supplier stability, integrated trauma portfolios, and long-term cooperation capability.
While multinational orthopedic companies continue to dominate premium hospital markets, distributor-oriented manufacturers with flexible cooperation models and broader orthopedic integration capability are becoming increasingly relevant in international distribution channels.
For distributors evaluating orthopedic trauma sourcing strategies, factors such as portfolio integration, instrument compatibility, OEM flexibility, and regulatory support remain central to long-term supplier selection.
CZMEDITECH provides a full range of orthopedic trauma solutions including intramedullary nails, external fixators, locking plates, and surgical instruments for global distributors.
Español
Русский
Français
English
العربية
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu











