Orthopaedic Supplier dik tak thlan dan — Indonesia Hospital Expo Insights
Thil thleng: Indonesia Int'l Hospital Expo 2025 neih turah hian | Ni: September 25–28, 2025 chhung khan | Booth: Hall 2-428-ah a awm a | Hmun: Indonesia Inkhawmpui Exhibition (ICE), Tangerang, Banten-ah neih a ni
1. Indonesia Hospital Expo 2025-a tel kan thlan chhan
Medical device industry-ah chuan rinna dik tak siam hi mimal inzawmna atanga tan a ni fo thin. Chu rinna chuan CZMEDITECH chu Indonesia International Hospital Expo 2025 -ah a tel ve ta a ni . Southeast Asia-a healthcare market lian ber pawl a nih avangin Indonesia hian orthopaedic lama thil thar siam leh accessibility inkara inzawmna pawimawh tak a ni.
Kan tum ber chu khawvel pum huapa CE leh ISO13485 standard nena inmil tha tak tak pholan a ni orthopedic implant leh surgical solution — China atanga precision engineering hmanga he bial puma damdawi in, distributor leh surgeon te chakna a pek theih dan te lantir a ni bawk.
2. Hall 2-428-a Kan Booth atanga thil pawimawh ber berte
-ah chuan Booth Hall 2-428 CZMEDITECH-a core system hrang hrang kan pholang a — spinal fixation leh anterior cervical plate atanga expandable titanium cages leh fibular intramedullary nails thlengin. Product tin hian himna, dinhmun nghet leh surgical efficiency lama kan ngaihtuahna a keng tel vek a ni.
Surgeon leh thil lei duh tam tak chuan kan device-te chu mimal takin an enfiah a, thread geometry, surface finishing, leh material durability te an sawiho a. An feedback — a tam zawkah chuan pawmpuina kawng awlsam tak, dik tak — chuan kut themthiamna tak tak chuan thupui aiin thu a la ring zawk tih min hriat nawn tir a ni.
3. Key Outcomes leh Market Insights te
Exhibition hian Indonesia, Thailand, Malaysia, leh Middle East atanga healthcare professional-te nena inbiakna awmze nei tak neih theihna kawng a hawng a ni. Tlawhtute chuan kan integrated manufacturing capabilities — R&D leh CNC machining atanga anodizing leh surface treatment thlengin an ngaihven hle.
Tunlai thil lei duhtute hian man chauh ni lovin , delivery rintlakna, certification transparency, leh after-sales responsiveness te pawh an ngai pawimawh tih kan hre ta a ni . Heng hriatnate hian Southeast Asia-a kan thawhhona chhunzawm zelna tur a kaihruai ang a, chutah chuan tualchhung thawhhona leh technical rintlakna chu a pawimawh tak zet a ni.
4. The Ultimate Guide: Orthopaedic Supplier dik tak thlan dan
Kum tam tak chhunga industry lama kan tawnhriat leh kumin exhibition atanga kan thil hriatte hmangin, orthopaedic supplier rintlak tak hriat theihna atana thu bul pawimawh pathum kan tarlang a ni:
Certification hmasa ber: CE, ISO13485, emaw FDA zawm leh zawm loh enfiah fo ang che — an ni hi product quality leh damlo himna lungphum an ni.
Brochure Beyond Ask: Thil siam chhuah dan te, material sourcing te, leh post-sale service te zawt rawh. Supplier rintlak tak takte chuan an hnathawh dan chungchangah an inhawng reng a ni.
Professionalism Evaluate: Mithiam dik takte chuan presentation aiin an kal a — i market mamawh ang zela technical suggestion an pe thin.
Pro Tip: Exhibition zawh hnu rei takah pawh inbiakpawhna chhunzawm zel supplier zawng rawh — consistency signals commitment.
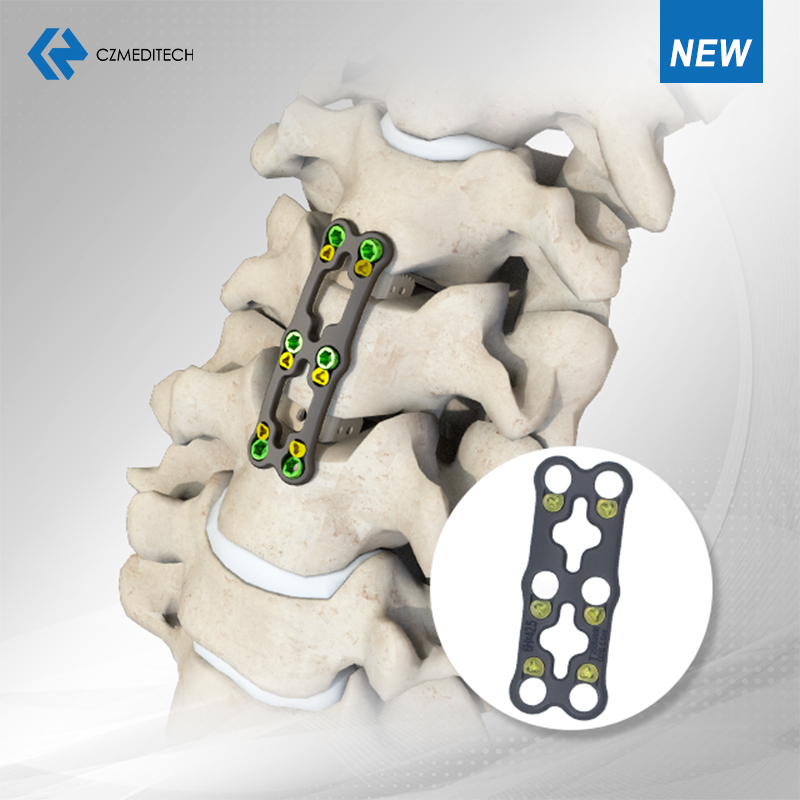
5. Thil siam chhuah langsar tak takte
Spinal Fixation System — Stabilization thar leh a inmilna dik tak.
Anterior Cervical Plate System — Fusion result tha ber tur atana engineer a ni.
PEEK Cage — Ruh thanna leh hun rei tak chhunga biocompatibility a tichak.
Fibular Intramedullary Nail — Trauma leh reconstructive application atana siam a ni.
![1 1]()
Cervical tihzauh theih Titanium Mesh Cage
Implant Material: Titanium Alloy A hman theih dan: Cervical corpectomy defects reconstruction Vertebral body tumor resection hnua siam thar leh Vertebral body fracture na tak, corporectomy ngai
![2 2]()
Titaium Mesh a ni
Implant Material: Commercially Pure Titanium emaw Titanium Alloy hman theih dan: Luruh chhiatna avanga cranioplasty Orbital wall fracture siam thar leh Mandibular siam thar Maxillofacial ruh chhiatna siam thar leh
![3 3]()
Cranial Clamps hmangin Luruh Lock a awm
Implant Material: Titanium Alloy A hman theih dan: Neurosurgical craniotomies-a ruh flap fixation Naupangte craniofacial surgery
![4 4]()
Distal Tibial Intramedullary Nail atanga chhuak a ni
Kan siam thar Distal Femoral Nail (DFN) leh Fibular Intramedullary Nail te hian lower limb fracture enkawlna atan solution hmasawn tak tak a pe a, soft tissue hliam tihtlem leh dam chakna a ngaih pawimawh hle.
6. Zawhna zawh fo thin (FAQ) .
Q1: I thil siamte hi international certified a ni em?
A1: Ni e, CZMEDITECH orthopedic implant zawng zawng hi CE leh ISO13485 certified vek a ni.
Q2: Orthopaedic solutions customized i pe em?
A2: A dik tak zet. Clinical mamawh ang zela dimension, angle, leh surface treatment-ah customization kan pe thin.
Q3: Engtin nge CZMEDITECH distributor ka nih theih ang?
A3: Tlawh mai rawh czmeditech.com/contact-us hmangin i details te chu submit rawh. Kan regional manager hian darkar 24 chhungin a rawn thleng ang.
7. Ngaihtuahna hnuhnung ber
Exhibition hi product showcase aiin a tam zawk — inzawmna moments a ni. Indonesia Hospital Expo 2025-ah khan surgeon-te hriatthiamna kan ngaithla a, nakin lawka kan thawhpui turte nen kut kan inthlak a, kan mission chu kan nemnghet leh a: China atanga orthopaedic innovation rintlak tak tak khawvel hriata rawn thlen.
Event tin hian CZMEDITECH hian khawvel pumah a zau zel a, surgical outcome tihhmasawn leh medical professional-te precision-engineered implants hmanga chakna pe turin a inpeih reng a ni.
![1080-2 1080-2 ah a awm]()
Thutawp leh Hmalam hun thlir dan
a hlawhtling taka kan telna hian Indonesia Int’l Hospital Expo 2025- a hmasawnna pawimawh tak a ni CZMEDITECH khawvel pum huapa hmasawnna leh Southeast Asia market strategy- .
Clinical mamawh lo danglam zel phuhruk thei tur kan siam chhuak chhunzawm zel ang a, kan siam chhunzawm zel orthopedic implant solutions ang a, exhibition atanga feedback hlu tak tak leh market insight kan hmuhte chu kan R&D, product design, leh regional marketing approach tihchangtlun nan kan hmang ang.
hrang hranga damdawi-in, distributor, leh medical professional-te nena thawhhona kan tihchak zel angin Indonesia leh a ṭhenawm ram , kan mission chu a chiang reng a ni — khawvel puma surgical outcome tichangtlungtu leh damlote nunphung tichangtlung thei tur orthopaedic solution him zawk, fing zawk, leh hlawk zawk pek chhuah.
Uni-C Standalone Cervical Cage ah hian a awm a
A hnunglam Cervical Screw System (Thar) 1. A rilru a hah lutuk chuan a rilru a buai em em a.
A hmalam Cervical Spine Fixation System a ni
Titanium Mesh hmanga siam a ni
Cranial clamps luruh Lock a ni
CERVICAL THIL TIH DAN THIL TIH DAN THIL THAWN THAWN THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL THIL
Distal Tibial Intramedullary Nail atanga chhuak a ni
Expert Fibular Intramedullary Nail hmanga siam a ni
Español
Русский
Français
English
العربية
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu