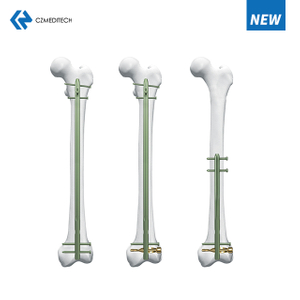
Femur (Femoral) Nail hman dan
Comminuted fractures a awm bawk
Segmental fractures a awm thin
Ruh hloh leh ruh tliak
Proximal leh distal fracture a awm thin
Union ni lo (nonunion) te
Subtrochanteric-a ruh tliak (subtrochanteric fractures) a awm
Intertrochanteric inkara inzawmna (intertrochanteric fractures) a awm
Femoral Intramedullary Nail hman dan tur.
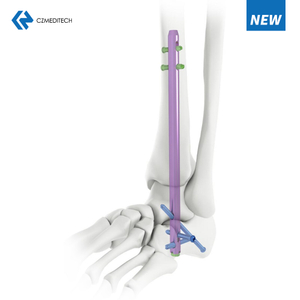
Femoral intramedullary nails (IM nails) hi surgical implants a ni a, chu chu ke ruh sei tak, femur ruh tliak enkawlna atana hman a ni. Chutianga tih dan tur chu nail chu femur intramedullary canal (the hollow center)-ah dahin, nail tawp lamah screw emaw, locking mechanism emaw hmangin a hmunah an nghet tlat a ni.
Femoral intramedullary nail hman dan tlangpui chu hetiang hi a ni:
Anesthesia: Damlo chu anesthesia pek a ni a, general emaw regional (spinal emaw epidural emaw) emaw a ni.
Incision: Surgeon chuan hip emaw knee joint bulah incision a siam a, chu chu a ruh tliakna hmun a zirin a ni.
Femur buatsaih dan: Surgeon chuan ruh chhungah intramedullary canal luh theihna turin kawngkhar a siam a.
Nail dah: Nail chu canal chhungah dahin, a ruh tliakna hmunah chuan hmasawn a ni.
Alignment: Surgeon chuan fluoroscopy (real-time X-ray) hmangin nail chu ruh nen a inmil tha em tih leh ruh tliak chu a tlahniam tawh tih a finfiah thin.
Fixation: Screw emaw locking mechanism hmangin ruh tawp lamah nail chu a hmuna nghet taka dah theih a ni.
Closure: Incision chu suture emaw staple emaw hmanga khar a, sterile dressing hnawih a ni.
Postoperative care-ah hian natna enkawl dan te, taksa lam enkawlna te, leh a dam dan enfiahna atana X-ray enkawl zui te a tel a ni.
Hriat tur pawimawh tak chu, step leh technique bikte chu femoral intramedullary nail hman chi hrang hrang a zirin a danglam thei a, chubakah surgeon duh dan leh tawnhriat a zirin a inang lo thei bawk. Procedure hi orthopaedic surgeon trained leh tawnhriat ngah chauhvin tih tur a ni.
Femur (Femoral) Nail tih loh tur
Damdawi thiam zirna, training leh professional rorelna rinchhan a ngai a, hmanraw leh enkawlna tha ber thlan a ngai a ni.
Hlawhchhamna hlauhawmna sang zawk lantir thei dinhmunte chu:
Active emaw rinhlelh latent infection emaw marked local inflammation emaw chu a natna hmun chhungah emaw, a chhehvelah emaw a awm.
Vascularity tihchhiat, chu chuan fracture emaw operative site emaw-a thisen supply tling a tikhawlo thei a ni.
Bone stock natna, infection emaw implantation hmaa tihchhiat, devices te support leh/ emaw fixation tha tawk thei lo.
Material sensitivity, documented emaw rinhlelh emaw.
Thau lutuk. Damlo thau lutuk emaw, thau lutuk emaw chuan implant-ah load a siam thei a, chu chuan device fixation a tichhe thei a, device ngei pawh a chhiat phah thei bawk.
Operation site chunga tissue coverage tling lo damlote.
Anatomical structures emaw physiological performance emaw tibuaitu tur implant hman dan.
Mental emaw neuromuscular disorder eng pawh, postoperative care-a fixation failure emaw complication emaw awm thei tur, pawm theih loh risk siam thei.
Surgery hlawkna awm thei tur tikhawlo thei tur medical emaw surgical condition dang.
Femoral Intramedullary Nail quality tha tak lei dan tur.
Femoral intramedullary nail quality tha tak lei i tum a nih chuan kaihhruaina tlangpui thenkhat chu hetiang hi a ni:
Damdawi hmanrua siamtu emaw, supplier emaw hmingthang tak chu zirchiang la, thlang rawh. Implant leh instrument tha tak tak siam chhuahna lama track record dik tak nei company zawng rawh.
Certification leh regulatory zawm leh zawm loh enfiah rawh. Thil siamtu emaw supplier emaw chuan FDA (US-ah), CE (EU-ah), emaw ISO (International Organization for Standardization) ang chi regulatory body atanga certification leh approval tul tak tak a dawng tih enfiah rawh.
Product specification te chu verify rawh. Femoral intramedullary nail hian a lian zawng, a pianzia leh a thil siam danah a mamawh ang specification a tlin em tih enfiah rawh.
Implant-a thil awmte hi han ngaihtuah teh. Locking mechanism, adjustable angle, leh anti-rotation capabilities ang chi feature zawng la, chu chuan damlo leh surgeon tan hlawkna dang a pe thei a ni.
Damdawi lam thiamte nen inrawn rawh. Thil lei hmain, i mamawh bik angin femoral intramedullary nail hi a inmil em tih hriat nan orthopedic surgeon thiam tak emaw, medical professional dang emaw i zawt hmasa phawt a pawimawh.
A dahna leh hman dan dik tak enfiah rawh. Implant i lei hnuah, surgery-a hman tur atana a dinhmun tha ber tura a awm theih nan a siamtu thupek angin dah leh khawih ngei ngei tur a ni.
CZMEDITECH chungchang
CZMEDITECH hi medical device company a ni a, orthopaedic implant leh instrument tha tak tak siam leh hralhna lama tui tak a ni a, intramedullary nail te pawh a tel. Company hian kum 14 chuang zet he industry-ah hian experience a nei tawh a, thil thar siam, quality leh customer service lama a inpekna avangin hriat hlawh tak a ni.
CZMEDITECH atanga intramedullary nail an lei hian customer te chuan quality leh safety lama international standard zawm thei thil, ISO 13485 leh CE certification te chu an beisei thei a ni. Company hian thil siamna lama technology hmasawn tak tak leh quality control process khauh tak hmangin thil siam zawng zawng chu quality sang ber a nih theih nan leh surgeon leh damlote mamawh a phuhruk theih nan a hmang a ni.
CZMEDITECH hian a thil siam tha tak tak bakah hian customer service tha tak a neih avangin hriat hlawh tak a ni bawk. Company hian sales representative tawnhriat ngah tak tak team an nei a, an thil lei chhung zawngin customer te hnenah kaihhruaina leh puihna an pe thei a ni. CZMEDITECH hian after-sales service kimchang tak a pe bawk a, technical support leh product training te pawh a pe bawk.
Español
Русский
Français
English
العربية
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu